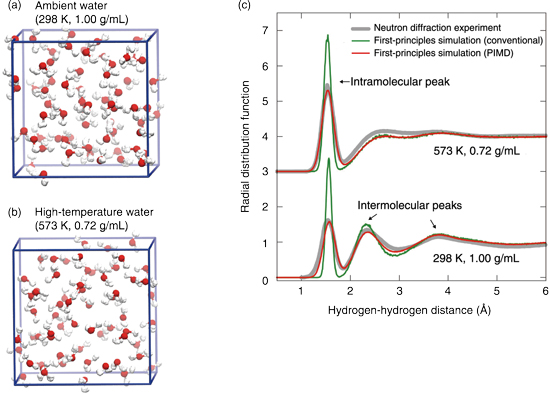
Fig.9-6 Structure and interatomic radial distribution function of water
Matter is composed of atoms, and molecular simulations that follow the collective motion of atoms can provide information on the properties of matter. Since atomic motion is determined by the behavior of electrons in each atom, it is necessary to clarify the behavior of electrons by a method called “first-principles calculation”, which is based on the quantum mechanics governing the behavior of electrons, and to calculate the atomic motion based on the results of this calculation-this process is called “first-principles simulation”. First-principles simulations are required to further calculate atomic motion based on the results of these calculations. First-principles simulations, which require large, high-performance computers, are revealing previously unknown microscopic behaviors of various materials. It was known that the conventional first-principles simulations cannot reproduce the experimental results for determining the structure of water, which is one of the most familiar substances. This is because the hydrogen atoms in water molecules are very light and are affected by quantum mechanics in the same way as electrons, and therefore, they exhibit “quantum fluctuation” (a state in which the position of an object can exist only stochastically). Such quantum fluctuations are not taken into account in the conventional first-principles simulations. If this effect can be accurately calculated using the first-principles simulations, it will be possible to evaluate more accurately various chemical reactions in water, including the reprocessing of nuclear fuel and dynamics of radiation elements in environments. In addition to the first-principles calculations of electrons, calculations dealing with the quantum fluctuations of nuclei are necessary for this purpose. Therefore, the Center for Computational Science & e-Systems has developed a calculation method that incorporates the quantum fluctuation of atomic nuclei and the software “PIMD” as a fundamental research technology in the field of nuclear energy. Using this technology, we performed the first-principles simulations of water. Fig.9-6 compares a conventional first-principles simulation of the radial distribution function (distribution of distances between atoms) and a first-principles simulation incorporating the quantum fluctuation using PIMD with the results of a neutron diffraction experiment (an experiment to analyze the structure of materials using neutron diffraction by crystals). The quantum fluctuation of hydrogen atoms is larger than that of hydrogen atoms. The larger the quantum fluctuation of the hydrogen atom, the lower and wider are the peak of the graph. While the conventional first-principles simulations deviate from experiments in some areas, the results obtained using PIMD are in good agreement with the experiments and are expected to enable more accurate evaluation of chemical reactions such as the transfer of hydrogen from a water molecule to another molecule.
PIMD is available as open-source software at the website of the Center for Computational Science and e-Systems (https://ccse.jaea.go.jp/software/PIMD/index.en.html) and is used by researchers in Japan and abroad. PIMD features a variety of PIMD is characterized by its ability to be combined with various first-principles codes to perform advanced first-principles simulations, such as calculations incorporating the quantum fluctuations of atomic nuclei, and is useful in research in the fields of physics, chemistry, and materials science.
(Motoyuki Shiga)