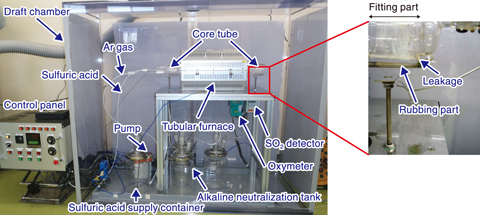
Fig.6-12 Designed corrosion test apparatus and location of SO2 leak
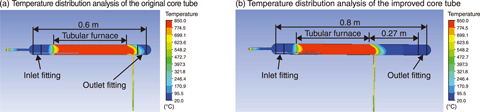
Fig.6-13 Effect of reducing the temperature of the fitting on the outlet side by optimizing the dimensions of the furnace tube
The IS process, a thermochemical hydrogen production method that is a potential heat-application technology of HTGRs, treats highly corrosive hydrogen iodide (HI), iodine (I2), sulfuric acid (H2SO4), and sulfur dioxide (SO2) at high temperatures. Any material involved in the process must have excellent heat resistance and corrosion resistance. In particular, the temperature inside the pressure vessel that decomposes sulfuric acid (i.e., the sulfuric acid decomposer) reaches the maximum temperature in the IS process of 850 ℃. Ceramics such as SiC have thus been considered for use as sulfuric acid decomposers. However, since SiC is expensive and making the sulfuric acid decomposer larger is difficult. As a result, it is also difficult to reduce the manufacturing cost because SiC must be jointed together. Heat-resistant and corrosion-resistant metallic materials that can replace SiC as the sulfuric acid decomposer are under development.
To commercialize said heat-resistant and corrosion-resistant metallic materials, the corrosion resistance of the developed material must be demonstrated in a sulfuric acid decomposition environment. A corrosion test apparatus was thus developed and demonstrated to provide excellent heat resistance and avoid corrosion for a long time (Fig.6-12). The apparatus consists of a control panel, a sulfuric acid supply container, a pump, a quartz glass core tube, a tubular furnace, and an alkali neutralization tank for neutralizing gasified SO2 by sulfuric acid. With the exception of the control panel, the supplies were stored in the draft chamber. By using argon as the carrier gas, gasified SO2 could be spread throughout the metal test piece installed in the core tube. For safety, several measurement devices were also installed to monitor the sulfuric acid solution, the amount of SO2 gas, the temperature distribution in the tubular furnace, and the oxygen concentration after sulfuric acid decomposition. If the temperature reaches 900 ℃ or SO2 leaks into the draft chamber, the system is automatically shut down. Since the sample is installed in the core tube for each test, both sides of the core tube have a fitted structure. In a trial run, liquid sulfuric acid flowed into the core tube; after a while, SO2 leaked from the fitting on the outlet side, triggering the automatic stop of the apparatus. This leakage was caused by the temperature rise of the rubbing part at the outlet of the core tube, which weakened the adhesive force of the fluorine grease applied to this part. Additionally, liquid sulfuric acid had pooled near the outlet-side fitting, which also contributed to the leakage of SO2 gas. To address this problem, thermo-fluid calculations were performed to optimize the dimensions of the core tube. The calculation results indicated that increasing the distance between the tubular furnace and the outlet-side fitting would reduce the temperature of the outlet fitting to room temperature; using the determined dimensions, a core tube that could be safely stored in the draft chamber was designed (Fig.6-13).
In fact, SO2 leakage does not occur in the improved core tube for a long period because the number of steps inside the core tube is minimized to eliminate liquid pools, and the core tube is lengthened to avoid temperature rise. Corrosion tests on various metallic materials using the designed apparatus are ongoing. Future work will include the development of a heat-resistant and corrosion-resistant metallic material for use as a sulfuric acid decomposer.
(Noriaki Hirota)