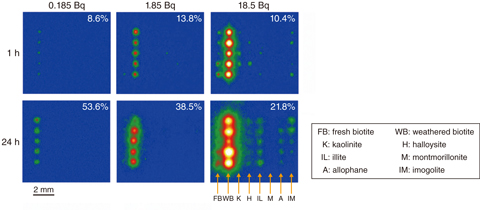
Fig.1-31 Read-out images of IPs with particles of various minerals adsorbing 137Cs from the solutions
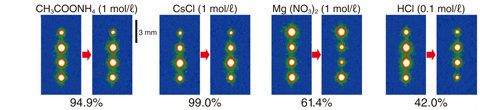
Fig.1-32 IP read-out images to show the desorption feature of 137Cs from WB using various chemical reagents
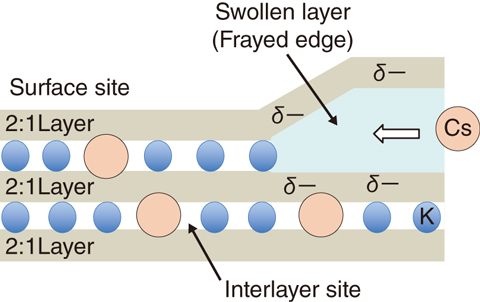
Fig.1-33 A model of radioactive-cesium adsorption into WB
After the accident at the TEPCO’s Fukushima Daiichi NPS in March 2011, many radioactive materials were dispersed in the environment and huge amounts of radioactive soil waste were generated by the decontamination project to reduce air-dose rate. Most of the radioactive cesium (Cs) in the waste was fixed in clay minerals. On the basis of this result, the development of stable interim-storage and volume-reduction methods for the waste is strongly desired. Thus, Cs sorption/desorption experiments for various clay minerals, considering actual contamination conditions in Fukushima, were conducted to elucidate the sorption mechanism of Cs into the clay. This was done using 137Cs radioisotope and autoradiography using imaging plates (IPs).
In this experiment, eight clay-mineral species with five particles were arranged on an acrylic substrate; then, aqueous solutions containing low concentration of 137Cs were dropped to cover all particles on the substrates. Fig.1-31 shows the read-out images of the IPs. Under any 137Cs concentration and immersion periods, the amount 137Cs adsorbed by weathered biotite (WB) collected from Fukushima was much higher than that of other minerals. Therefore, when WB was present in the soils, Cs is considered to be selectively adsorbed onto this mineral.
Moreover, desorption experiments were conducted using the Cs-adsorbed particles of WB and various chemical reagents for elution. Consequently, the elution of 137Cs was observed only when acid sufficiently strong to dissolve clay minerals was used as an eluent (Fig.1-32). This result suggests that Cs is strongly fixed to interlayer sites of WB (Fig.1-33) and that Cs, once adsorbed on WB, hardly leaches out into the environment.
This study revealed that WB is an important material for understanding radioactive contamination in Fukushima soils. For example, the existence of WB will dominate the fixing and leaching behaviors of radioactive materials. These results are expected to lead to the suggestion of effective reduction and safe interim-storage methods for radioactive waste soils.
This study is a part of the result of contract research “Elucidation of adsorption and transition mechanism of radioactive cesium in soil particle” commissioned by the University of Tokyo.